Meteorite Classes
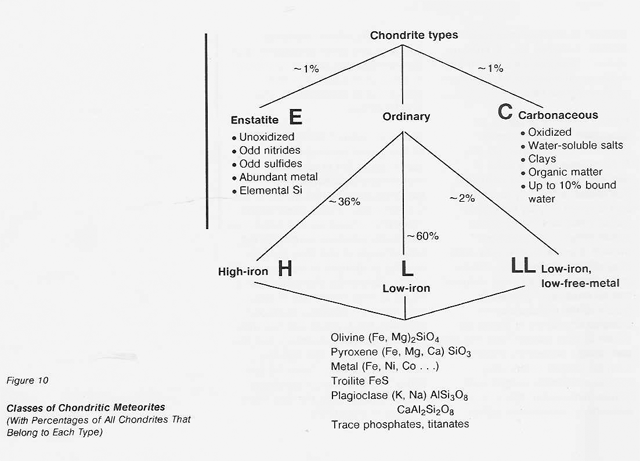
Figure 9 shows the general scheme for classification of meteorites. This scheme unifies the more than 30 known classes under the three principal headings of stones, stony- irons, and irons. Primitive solid material, in which the major rock- forming elements have about the same relative abundances as in the Sun, accounts for the subset of stony meteorites called chondrites. All other classes of meteorites are the results of melting and density-dependent geochemical differentiation of primitive material. Chondrites.fall on Earth far more often than all these other meteorite types combined. The prospect of mining differentiated asteroids is not encouraging. Volatiles are probably rare or absent, and free metals will probably have drained into a massive monolithic core. Conversely, chondritic asteroids, both those with high volatile content and those with high free-metal content, are attractive targets.
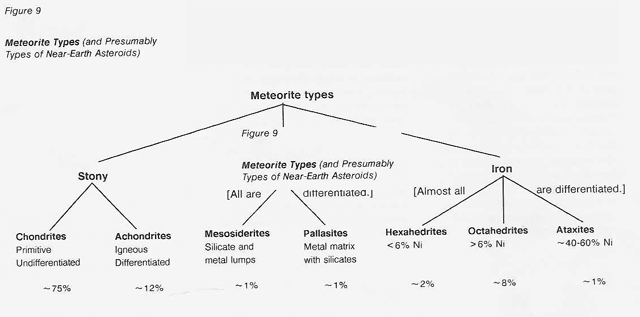
The various classes of chondrites differ greatly in mineralogy, oxidation state, and volatile content (fig. 10) as a consequence of having formed at different temperatures. The content (by weight) of volatile-rich, low- temperature carbonaceous (C1) chondrites is up to 20 percent water chemically bound in clay minerals, up to 6 percent organic matter, and up to 11 percent magnetite. Nitrogen is present in the organic matter, and sulfur may be found as sulfides, elemental sulfur, and water-soluble sulfates. Carbonates and halides are abundant. All members of the C1 subtype of carbonaceous chondrites are very easily crushed; so are the members of the C2 subtype. But the crushing strength of all other meteorite classes, including the C3 subtype, varies.* Carbonaceous chondrites make up on the order of 1 percent of all meteorite falls.
Equally rare, the enstatite (E) chondrites display markedly different compositions. All the E chondrites are in a state of extraordinary chemical reduction. Iron oxides are wholly absent and iron is found only as the sulfide troilite (FeS) and in iron-nickel- cobalt alloys. The dominant mineral is enstatite, the very pure magnesium orthosilicate. These meteorites are so strongly reduced that as much as 1 percent by weight of the metal phase in enstatite chondrites is elemental silicon in solid solution with the iron and nickel. Accessory materials such as calcium sulfide (the mineral oldhamite), magnesium sulfide (niningerite), titanium nitride (osbornite), manganese sulfide (alabandite), silicon oxynitride (sinoite), and even potassium- and titanium-bearing sulfides are found in the E chondrites or in their differentiated counterparts, the E achondrites.
However, more than 95 percent of the chondrites that fall on Earth (about 3/4 of all known meteorites) lie between the extremes represented by the E and C chondrites. These intermediate "ordinary" chondrites are subdivided into three groups according to the total amount of iron they contain and the proportion of that iron (and of the siderophilic elements) that is found as free metal: the H group with high iron content (much of it metallic), the L group with low iron content (less of it metallic), and the LL group with low iron and low free metal content. Table 15 and figure 11 show the compositional relationships among the five major classes of chondrites.

*Michaei Lipschutz, personal communication with the editor
Next
|
Curator: Al Globus If you find any errors on this page contact Al Globus. |
 |
This site was hosted by the NASA Ames Research Center from 1994-2018 and is now hosted by:
