|
|
 |
Group size
Whole class
Time required
15 minutes
Materials
National standards (5-8) addressed
Student Objectives
Student will:
In this activity, condensation nuclei are provided by a wisp of smoke. In the atmosphere, water droplets condense around dust particles.
This demonstration shows that rapid decrease in pressure will lower the temperature of a gas (Gay-Lussac's Law).
Advance Preparation Time
Get materials
Teacher tips
Clouds, rain, wind and other weather phenomena not only
desirable but may be necessary in orbital space settlements.
Beautiful clouds may also be visually appealing and give a sense of
outdoors to the colonists. From studies in biosphere 2 we now know
that wind is essential for trees. Wind causes the branches of trees
to be strong, and grow in an orderly fashion. This activity will
present students some ideas on how clouds could be formed in space
settlements. Very moist air would be pressurized and be allowed to
expand into slightly dusty or smoky air. When moist air expands,
clouds will form with dust and smoke particles becoming
"seeds".
Step 1
Blow up the balloon once to stretch it. Deflate it and cut off
stem. Stretch the balloon over the mouth of the jar to make sure it
fits tightly. Then remove it, but keep it ready to replace
quickly.
Step 2
Put about a teaspoon of water in the bottom of the jar.
Step 3
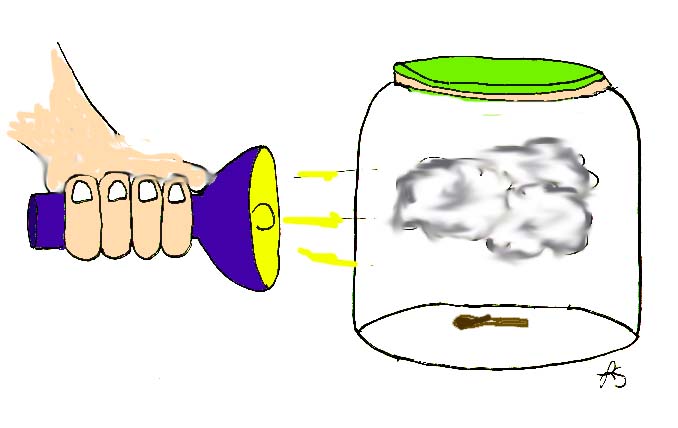
Drop a burning match into the jar to provide a wisp of smoke.
Immediately cover the mouth of the jar with the balloon. Fasten the
balloon in place with the rubber band. The air in the jar has been
"seeded".
Step 4
Push down on the center of the balloon with your knuckles. This
compresses the air in the jar and increases the pressure.
Step 5
Grasp the balloon in the middle between your thumb and forefinger,
and pull up on it quickly. The sudden decrease in pressure cools
the air. With a little luck a "cloud" will form in the jar. If you
push down and pull up again quickly, the cloud will disappear and
reappear again and again.
A beam of light directed through the jar and a dark background will make the effect more dramatic.
Enrichment Activities
If you have access to lab equipment, a better way of doing this
demonstration is by using a bicycle hand pump or compressed air.
Instead of a jar, use a bottle with a one hole rubber stopper.
After throwing the match into bottle, immediately raise the
pressure inside the bottle by pumping air into the bottle through
the hole of the rubber stopper. Let the air expand, by lifting the
bicycle pump out of the rubber stopper hole. Immediately shine the
flashlight to the bottle to show the "cloud" that formed inside the
bottle. Repeat to show the students that this is a reversible
process.
Illustration by Leyla Sezen.
|
|
 |
Author: Tugrul Sezen
|
Curator: Al Globus If you find any errors on this page contact Al Globus. |
 |
This site was hosted by the NASA Ames Research Center from 1994-2018 and is now hosted by:
